EX-99.2
Published on March 9, 2026
| BREAKTHROUGH ANTIBODIES FOR OBESITY AND CARDIOMETABOLIC DISEASES CORPORATE PRESENTATION March 2026 |
| Certain statements in this presentation constitute "forward -looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995 , as amended. Words such as "may," "might," "will," "should," "believe," "expect," "anticipate," "estimate," "continue," "predict," "forecast," "project," "plan," "intend" or similar expressions, or statements regarding intent, belief, or current expectations, are forward -looking statements. These forward -looking statements are based upon current estimates and includes statements regarding near term catalysts. While iBio, Inc., a Delaware corporation (including its consolidated subsidiaries, “iBio,” the “Company,” “we,” “us” or “our”) believes these forward - looking statements are reasonable, undue reliance should not be placed on any such forward -looking statements, which are based on information available to us on the date of this presentation. These forward -looking statements are subject to various risks and uncertainties, many of which are difficult to predict that could cause actual results to differ materially from current expectations and assumptions from those set forth or implied by any forward -looking statements. Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to obtain regulatory approvals for commercialization of its product candidates, or to comply with ongoing regulatory requirements, regulatory limitations relating to its ability to promote or commercialize its product candidates for specific indications, acceptance of its product candidates in the marketplace and the successful development, marketing or sale of products, its ability to attain license agreements, the continued maintenance and growth of its patent estate, its ability to establish and maintain collaborations, its ability to obtain or maintain the capital or grants necessary to fund its research and development activities, competition, its ability to retain its key employees or maintain its Nasdaq Stock Market listing, and the other factors discussed in the Company’s most recent Annual Report on Form 10 - K and the Company’s subsequent filings with the SEC, including subsequent periodic reports on Forms 10 - Q and 8 - K. The information in this presentation is provided only as of today, and we undertake no obligation to update any forward -looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law. Disclaimer. This presentation has been prepared by the Company solely for informational purposes. Certain of the information included herein was obtained from various sources, including certain third parties, and has not been independently verified by the Company. By viewing or accessing the information contained in this presentation, you hereby acknowledge and agree that no representations, warranties, or undertakings, express or implied, are made by the Company or any of its directors, shareholders, employees, agents, affiliates, advisors, or representatives as to, and no reliance should be placed on the truth, accuracy, fairness, completeness, or reasonableness of the information or opinions presented or contained in, and omission from, this presentation. Neither the Company nor any of its directors, employees, agents, affiliates, advisors, or representatives shall be responsible or liable whatsoever (in negligence or otherwise) for any loss, howsoever arising from any information presented or contained in this presentation or otherwise arising in connection with the presentation, except to the extent required by applicable law. This presentation includes statistical and other industry and market data that we obtained from industry publications and research, surveys, and studies conducted by third parties, and our own estimates of potential market opportunities. All of the market data used in this presentation involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such data. Industry publications and third - party research, surveys and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee, and we have not independently verified, the accuracy or completeness of such information. Our estimates of the potential market opportunities for our product candidates include several key assumptions based on our industry knowledge, industry publications, third -party research, and other surveys, which may be based on a small sample size and may fail to accurately reflect market opportunities. While we believe that our internal assumptions are reasonable, no independent source has verified such assumptions. Forward looking statements 2 |
| KFF Health Tracking Poll May 2024: The Public’s Use and Views of GLP-1 Drugs, KFF, May 10, 2025 3 Revolution Sparked a New Era in Obesity Treatment Incretin Class Agonists Have Revolutionized Obesity Treatment >10% of American adults have taken a GLP-1 1 Interventional weight loss previously only achievable via surgery Evolution Will Define Its Future Attention is Shifting to Therapies That Build on That Foundation Durability of weight loss Lean mass preservation and fat-specific weight loss Improved tolerability and convenience |
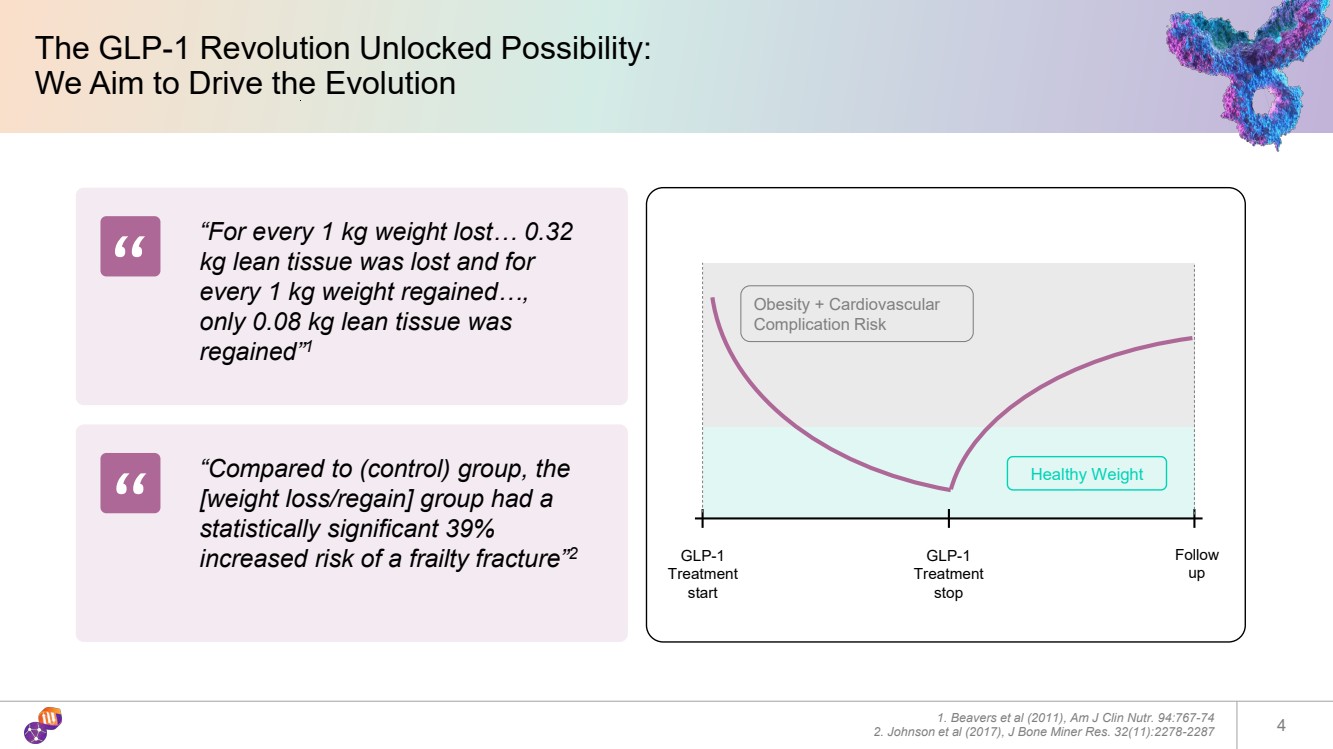
| The GLP-1 Revolution Unlocked Possibility: We Aim to Drive the Evolution 1. Beavers et al (2011), Am J Clin Nutr. 94:767-74 2. Johnson et al (2017), J Bone Miner Res. 32(11):2278-2287 4 GLP-1 Treatment start GLP-1 Treatment stop Follow up Obesity + Cardiovascular Complication Risk Healthy Weight “For every 1 kg weight lost… 0.32 kg lean tissue was lost and for every 1 kg weight regained…, only 0.08 kg lean tissue was regained”1 “Compared to (control) group, the [weight loss/regain] group had a statistically significant 39% increased risk of a frailty fracture”2 “ “ |
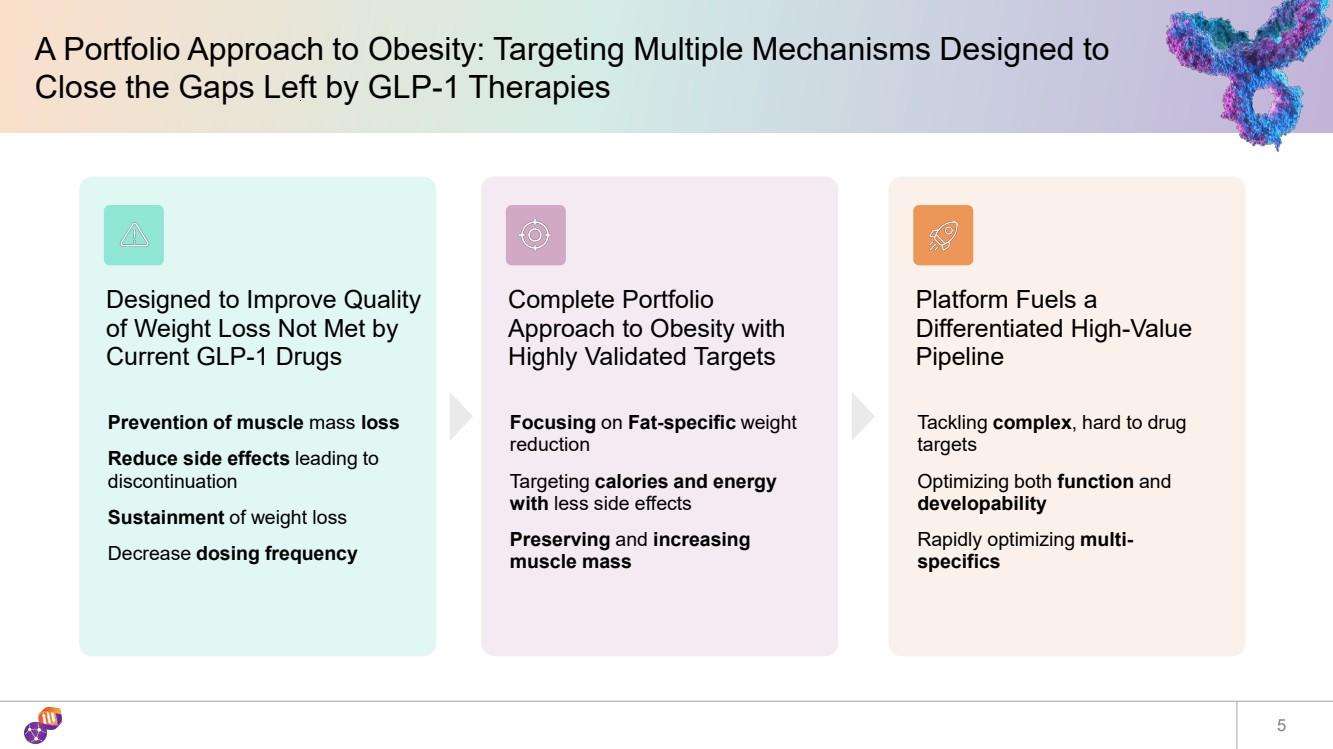
| A Portfolio Approach to Obesity: Targeting Multiple Mechanisms Designed to Close the Gaps Left by GLP-1 Therapies 5 Designed to Improve Quality of Weight Loss Not Met by Current GLP-1 Drugs Prevention of muscle mass loss Reduce side effects leading to discontinuation Sustainment of weight loss Decrease dosing frequency Complete Portfolio Approach to Obesity with Highly Validated Targets Focusing on Fat-specific weight reduction Targeting calories and energy with less side effects Preserving and increasing muscle mass Platform Fuels a Differentiated High-Value Pipeline Tackling complex, hard to drug targets Optimizing both function and developability Rapidly optimizing multi-specifics |
| Next Generation Antibodies for Obesity Targeting Key Gaps in Current Care Corporate Highlights Lead Programs ▪ IBIO-610: Activin E antibody ▪ Myostatin x Activin A: Bispecific Antibody Pipeline ▪ 3 early-stage high novelty programs and 2 partnered programs ▪ Discovery to development candidate in as little as 7 months ▪ AI engine delivers precisely targeted antibodies with promising developability ▪ IBIO-600: Long-acting myostatin antibody Anticipated Near Term Catalysts * Funding Dependent 6 Myostatin x Activin A IND equivalent filing expected in 1H 2027 IBIO-610 IND equivalent filing expected in 2H 2026 IBIO-610 Phase 1 expected to be initiated in 1H 2027* IBIO-600 IND equivalent filing expected, and Phase 1 initiated in 1H 2026 |
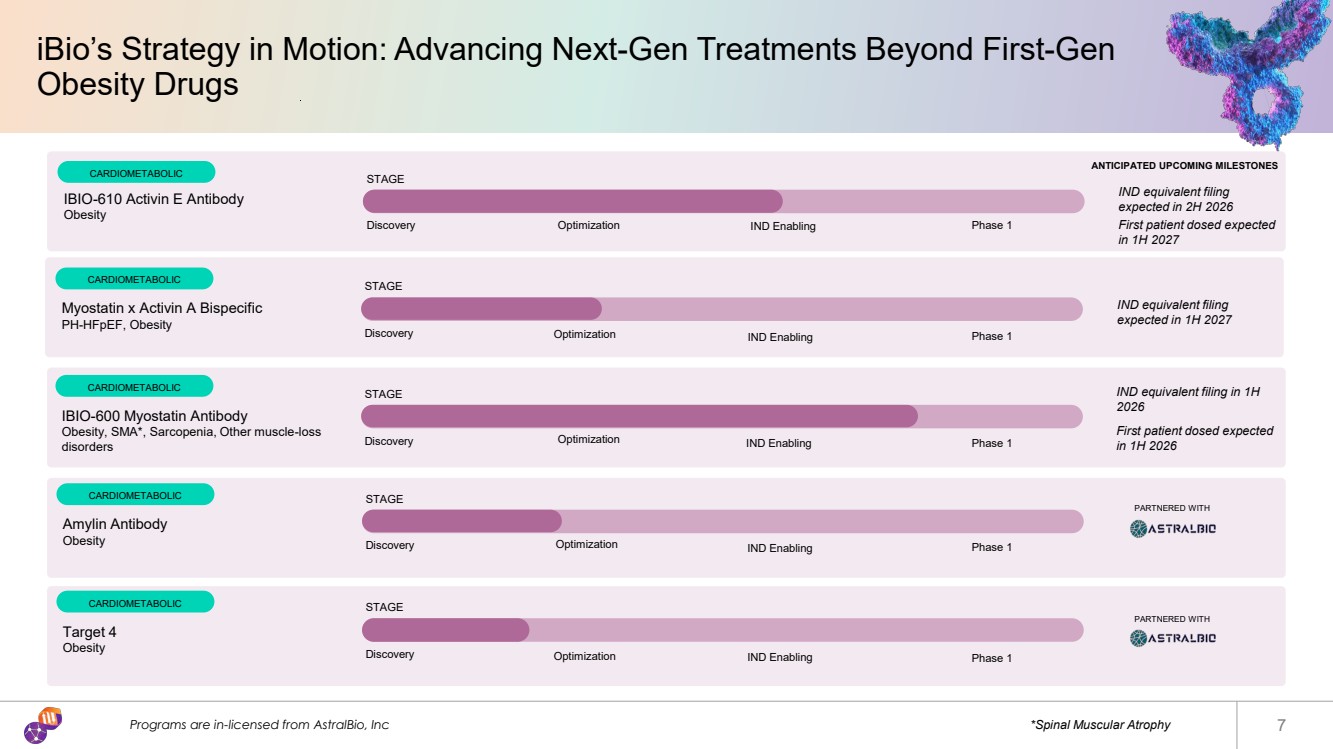
| iBio’s Strategy in Motion: Advancing Next-Gen Treatments Beyond First-Gen Obesity Drugs Programs are in-licensed from AstralBio, Inc *Spinal Muscular Atrophy 7 CARDIOMETABOLIC STAGE Discovery Optimization IND Enabling Phase 1 Myostatin x Activin A Bispecific PH-HFpEF, Obesity CARDIOMETABOLIC STAGE Discovery Optimization IND Enabling Phase 1 IBIO-600 Myostatin Antibody Obesity, SMA*, Sarcopenia, Other muscle-loss disorders CARDIOMETABOLIC STAGE Discovery Optimization IND Enabling Phase 1 Amylin Antibody Obesity CARDIOMETABOLIC STAGE Discovery Optimization IND Enabling Phase 1 Target 4 Obesity CARDIOMETABOLIC STAGE Discovery Optimization IND Enabling Phase 1 IBIO-610 Activin E Antibody Obesity IND equivalent filing expected in 2H 2026 First patient dosed expected in 1H 2027 IND equivalent filing expected in 1H 2027 IND equivalent filing in 1H 2026 First patient dosed expected in 1H 2026 PARTNERED WITH PARTNERED WITH ANTICIPATED UPCOMING MILESTONES |
| IBIO-610 Activin E Antibody |
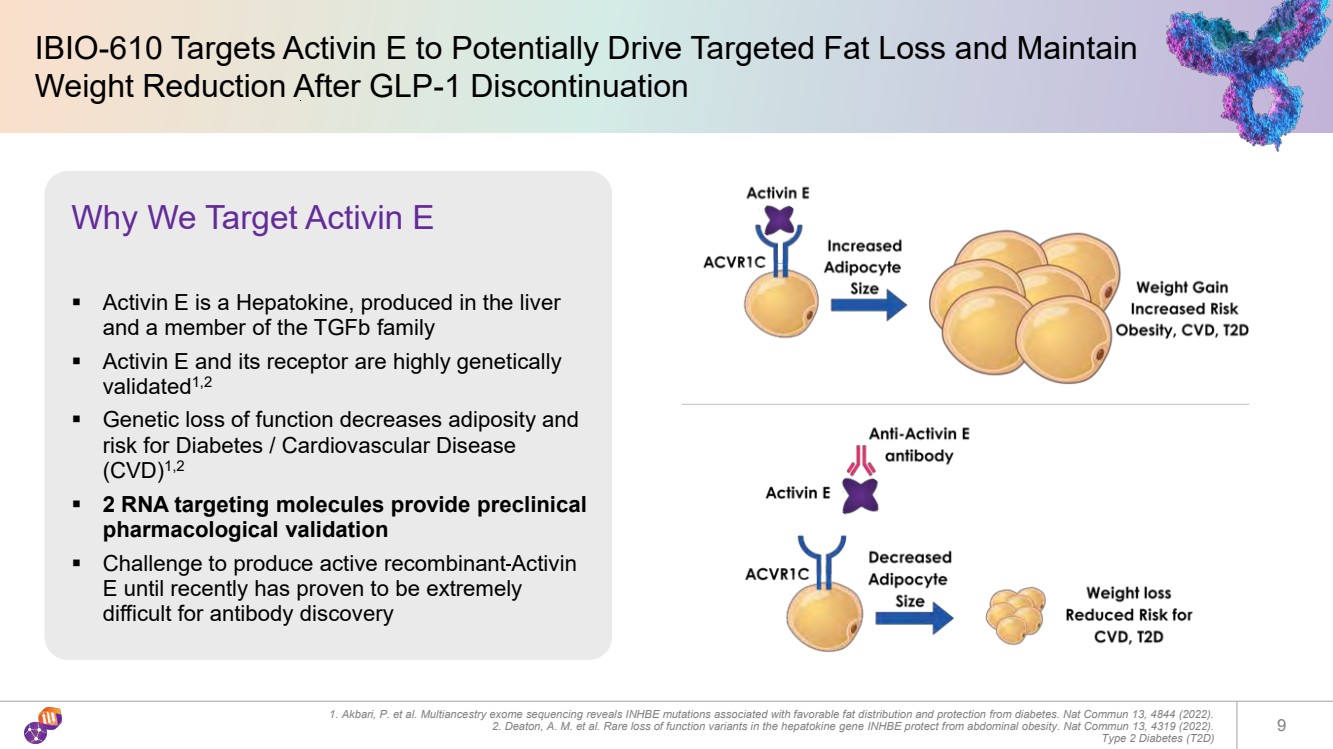
| IBIO-610 Targets Activin E to Potentially Drive Targeted Fat Loss and Maintain Weight Reduction After GLP-1 Discontinuation 1. Akbari, P. et al. Multiancestry exome sequencing reveals INHBE mutations associated with favorable fat distribution and protection from diabetes. Nat Commun 13, 4844 (2022). 2. Deaton, A. M. et al. Rare loss of function variants in the hepatokine gene INHBE protect from abdominal obesity. Nat Commun 13, 4319 (2022). Type 2 Diabetes (T2D) 9 Why We Target Activin E ▪ Activin E is a Hepatokine, produced in the liver and a member of the TGFb family ▪ Activin E and its receptor are highly genetically validated1,2 ▪ Genetic loss of function decreases adiposity and risk for Diabetes / Cardiovascular Disease (CVD)1,2 ▪ 2 RNA targeting molecules provide preclinical pharmacological validation ▪ Challenge to produce active recombinant Activin E until recently has proven to be extremely difficult for antibody discovery |
| IBIO-610 as a Potential First-in-Class Antibody Targeting Activin E 10 Potential Class-Leading Pathway Targeting Antagonist antibody offers potential for greater Activin E inhibition than siRNA-based knockdown approaches Fat Specific Weight Loss Weight loss observed in pre-clinical studies with no impact on lean mass Synergistic to GLP-1 Receptor Agonists Synergistic weight loss with appetite reducing drugs like GLP-1 or Amylin observed in pre-clinical studies. Weight Lowering and Maintenance Therapy Stand-alone weight loss intervention and weight loss maintenance post GLP-1 or Amylin treatment Optimized for Manufacturability Optimized for high expression and stability, enabling efficient production within a mature, globally scalable antibody manufacturing infrastructure |
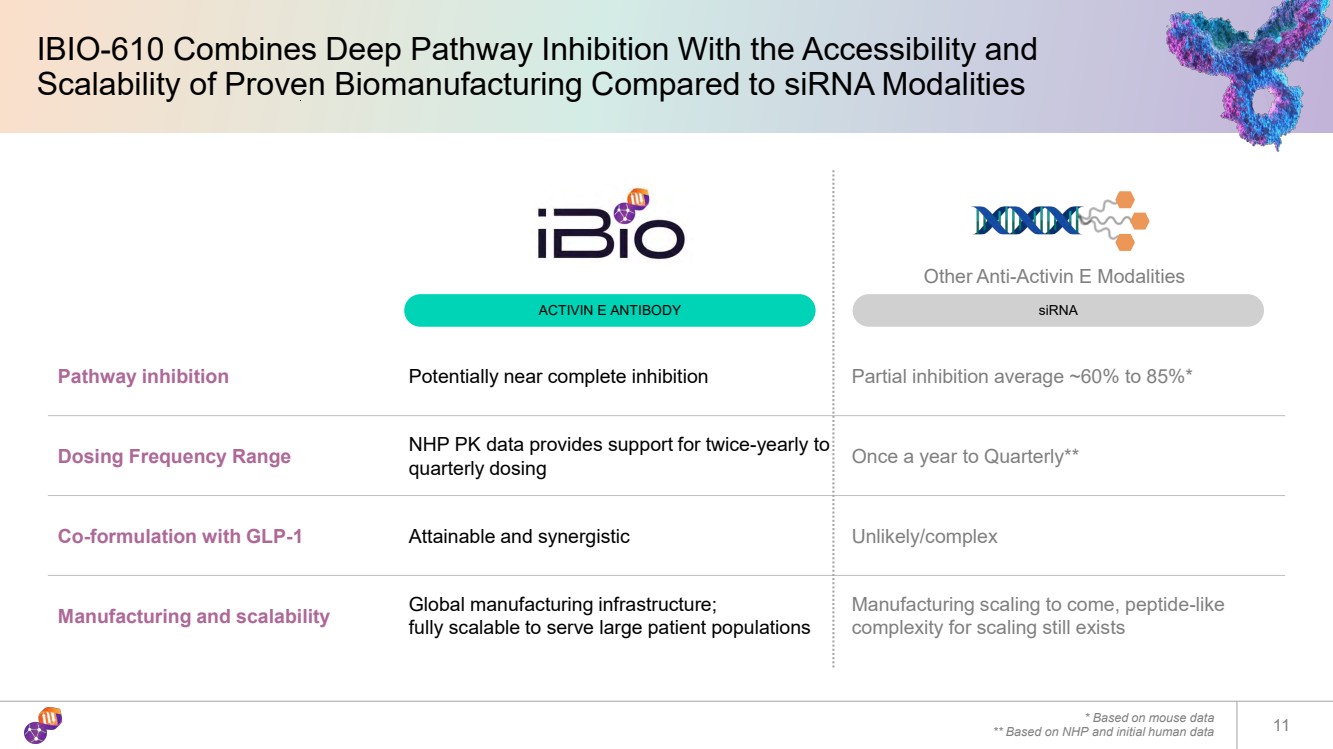
| IBIO-610 Combines Deep Pathway Inhibition With the Accessibility and Scalability of Proven Biomanufacturing Compared to siRNA Modalities * Based on mouse data ** Based on NHP and initial human data 11 Pathway inhibition Potentially near complete inhibition Partial inhibition average ~60% to 85%* Dosing Frequency Range NHP PK data provides support for twice-yearly to quarterly dosing Once a year to Quarterly** Co-formulation with GLP-1 Attainable and synergistic Unlikely/complex Manufacturing and scalability Global manufacturing infrastructure; fully scalable to serve large patient populations Manufacturing scaling to come, peptide-like complexity for scaling still exists Other Anti-Activin E Modalities ACTIVIN E ANTIBODY siRNA |
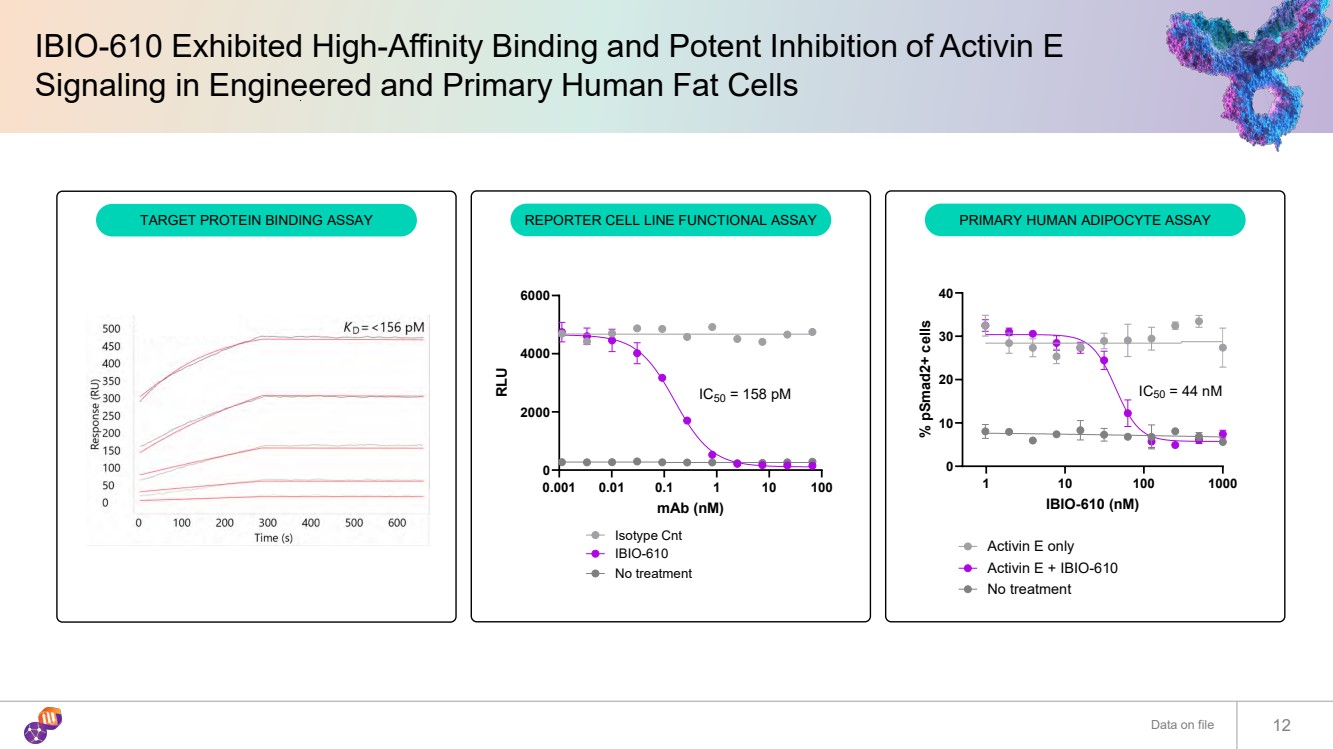
| IBIO-610 Exhibited High-Affinity Binding and Potent Inhibition of Activin E Signaling in Engineered and Primary Human Fat Cells Data on file 12 0.001 0.01 0.1 1 10 100 0 2000 4000 6000 mAb (nM) RLU Isotype Cnt IBIO-610 No treatment IC50 = 158 pM 1 10 100 1000 0 10 20 30 40 IBIO-610 (nM) % pSmad2+ cells No treatment Activin E only Activin E + IBIO-610 IC50 = 44 nM TARGET PROTEIN BINDING ASSAY REPORTER CELL LINE FUNCTIONAL ASSAY PRIMARY HUMAN ADIPOCYTE ASSAY |
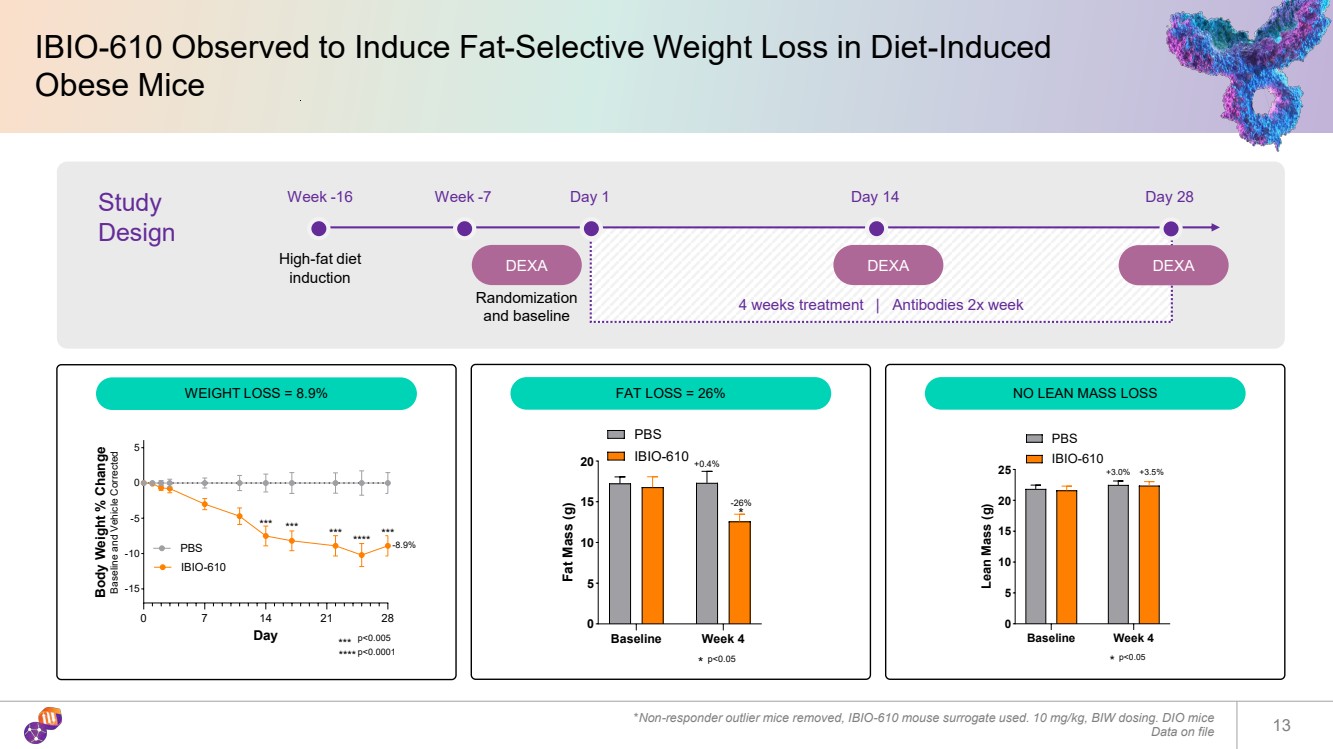
| IBIO-610 Observed to Induce Fat-Selective Weight Loss in Diet-Induced Obese Mice *Non-responder outlier mice removed, IBIO-610 mouse surrogate used. 10 mg/kg, BIW dosing. DIO mice Data on file 13 0 7 14 21 28 -15 -10 -5 0 5 Day Body Weight % Change Baseline and Vehicle Corrected PBS IBIO-610 -8.9% *** *** *** *** **** p<0.0001 p<0.005 *** **** Baseline Week 4 0 5 10 15 20 Fat Mass (g) * p<0.05 * +0.4% -26% PBS IBIO-610 Baseline Week 4 0 5 10 15 20 25 Lean Mass (g) * p<0.05 +3.0% +3.5% PBS IBIO-610 Study Design Week -16 High-fat diet induction Week -7 DEXA Day 1 DEXA Day 14 DEXA Day 28 Randomization and baseline 4 weeks treatment | Antibodies 2x week WEIGHT LOSS = 8.9% FAT LOSS = 26% NO LEAN MASS LOSS |
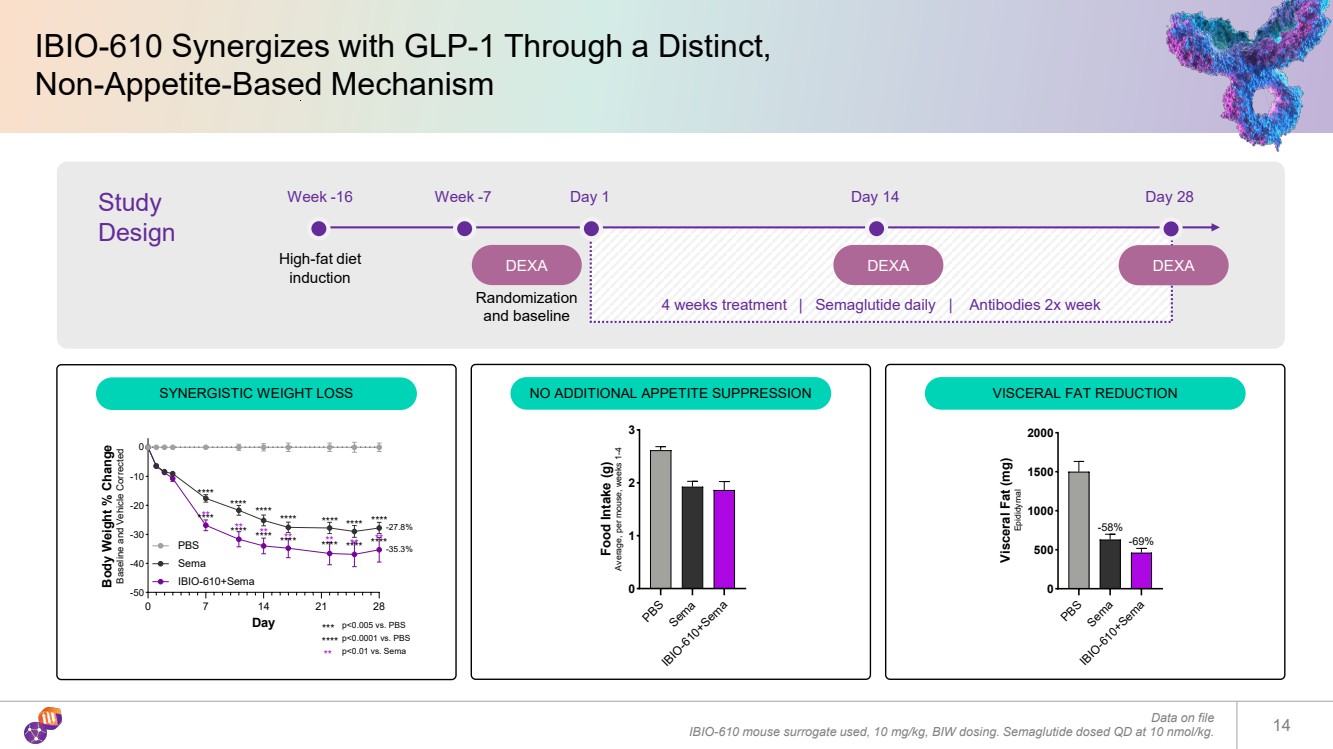
| IBIO-610 Synergizes with GLP-1 Through a Distinct, Non-Appetite-Based Mechanism Data on file IBIO-610 mouse surrogate used, 10 mg/kg, BIW dosing. Semaglutide dosed QD at 10 nmol/kg. 14 Study Design Week -16 High-fat diet induction Week -7 DEXA Day 1 DEXA Day 14 DEXA Day 28 Randomization and baseline 4 weeks treatment | Semaglutide daily | Antibodies 2x week SYNERGISTIC WEIGHT LOSS NO ADDITIONAL APPETITE SUPPRESSION VISCERAL FAT REDUCTION 0 7 14 21 28 -50 -40 -30 -20 -10 0 Day Body Weight % Change Baseline and Vehicle Corrected PBS Sema IBIO-610+Sema -27.8% -35.3% p<0.0001 vs. PBS p<0.005 vs. PBS p<0.01 vs. Sema **** *** ** **** ** **** ** **** **** ** **** ** **** ** ** **** ** **** **** **** **** **** **** **** PBSSema IBIO-610+Sema 0 1 2 3 Food Intake (g) Average, per mouse, weeks 1-4 PBSSema IBIO-610+Sema 0 500 1000 1500 2000 Visceral Fat (mg) Epididymal -58% -69% |
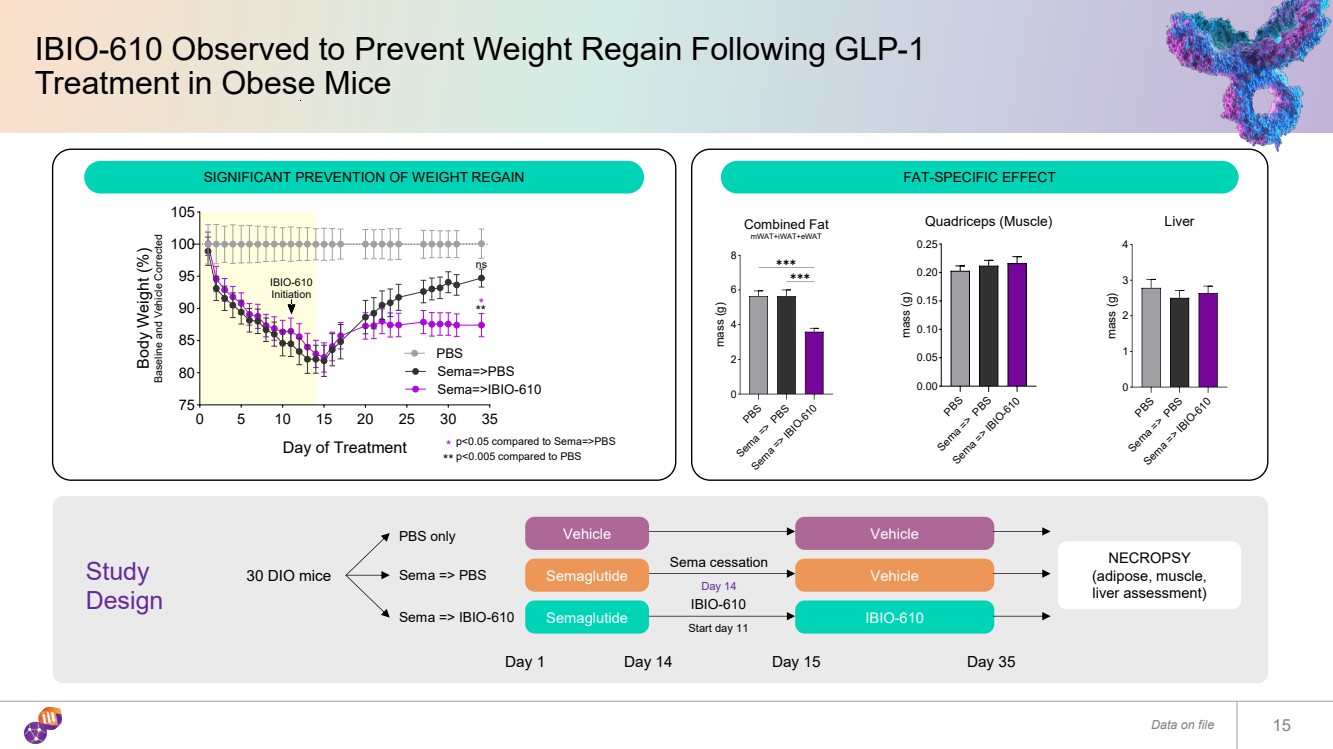
| IBIO-610 Observed to Prevent Weight Regain Following GLP-1 Treatment in Obese Mice Data on file 15 Fat-Specific Effect 0 5 10 15 20 25 30 35 75 80 85 90 95 100 105 Day of Treatment Body Weight (%) Baseline and Vehicle Corrected PBS Sema=>PBS Sema=>IBIO-610 IBIO-610 Initiation ns *** ** * p<0.05 compared to Sema=>PBS p<0.005 compared to PBS PBS Sema => PBS Sema => IBIO-610 0 2 4 6 8 Combined Fat mWAT+iWAT+eWAT mass (g) ✱✱✱ ✱✱✱ PBS Sema => PBS Sema => IBIO-610 0.00 0.05 0.10 0.15 0.20 0.25Quadriceps (Muscle) mass (g) PBS Sema => PBS Sema => IBIO-610 0 1 2 3 4 Liver mass (g) Vehicle Semaglutide Semaglutide 30 DIO mice PBS only Sema => PBS Sema => IBIO-610 Day 1 Sema cessation Day 14 IBIO-610 Start day 11 Day 14 Vehicle Vehicle IBIO-610 Day 15 Day 35 NECROPSY (adipose, muscle, liver assessment) SIGNIFICANT PREVENTION OF WEIGHT REGAIN FAT-SPECIFIC EFFECT Study Design |
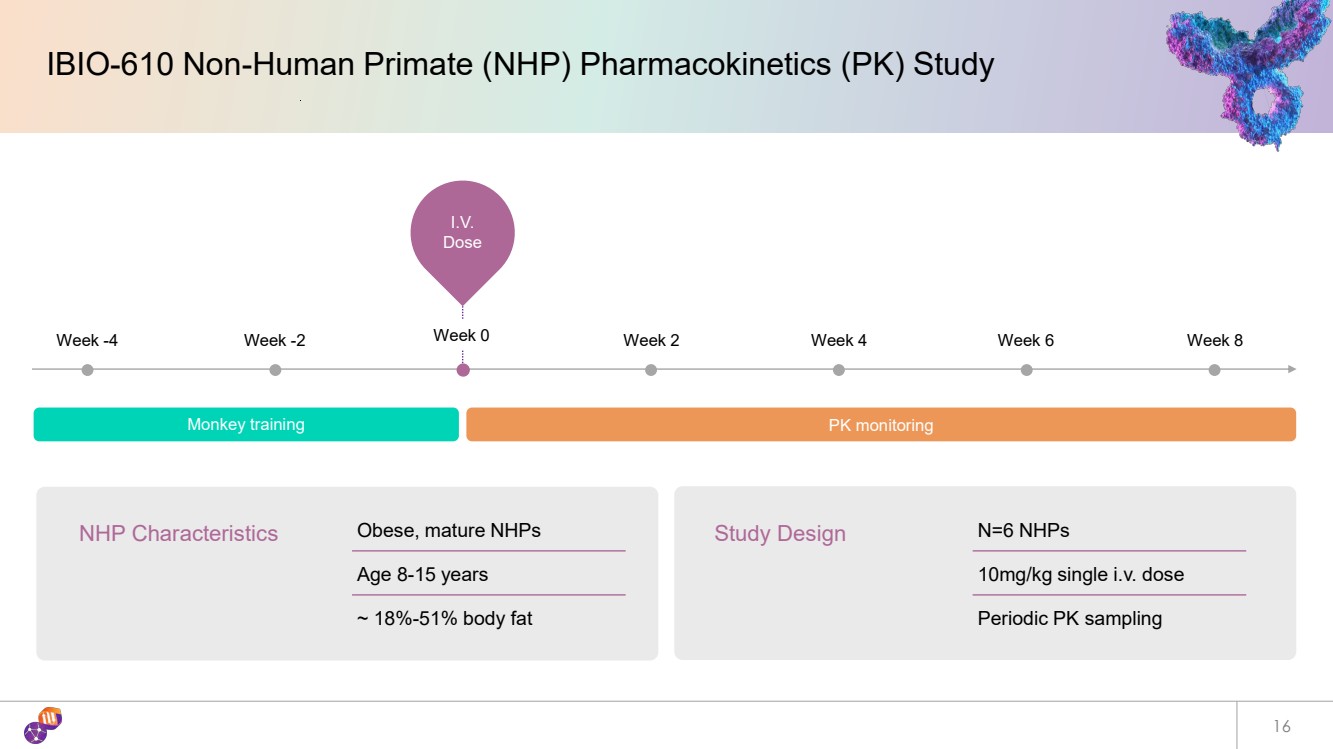
| 16 IBIO-610 Non-Human Primate (NHP) Pharmacokinetics (PK) Study I.V. Dose Week -4 Week -2 Week 0 Week 2 Week 4 Week 6 Week 8 Monkey training PK monitoring NHP Characteristics Obese, mature NHPs Age 8-15 years ~ 18%-51% body fat Study Design N=6 NHPs 10mg/kg single i.v. dose Periodic PK sampling |
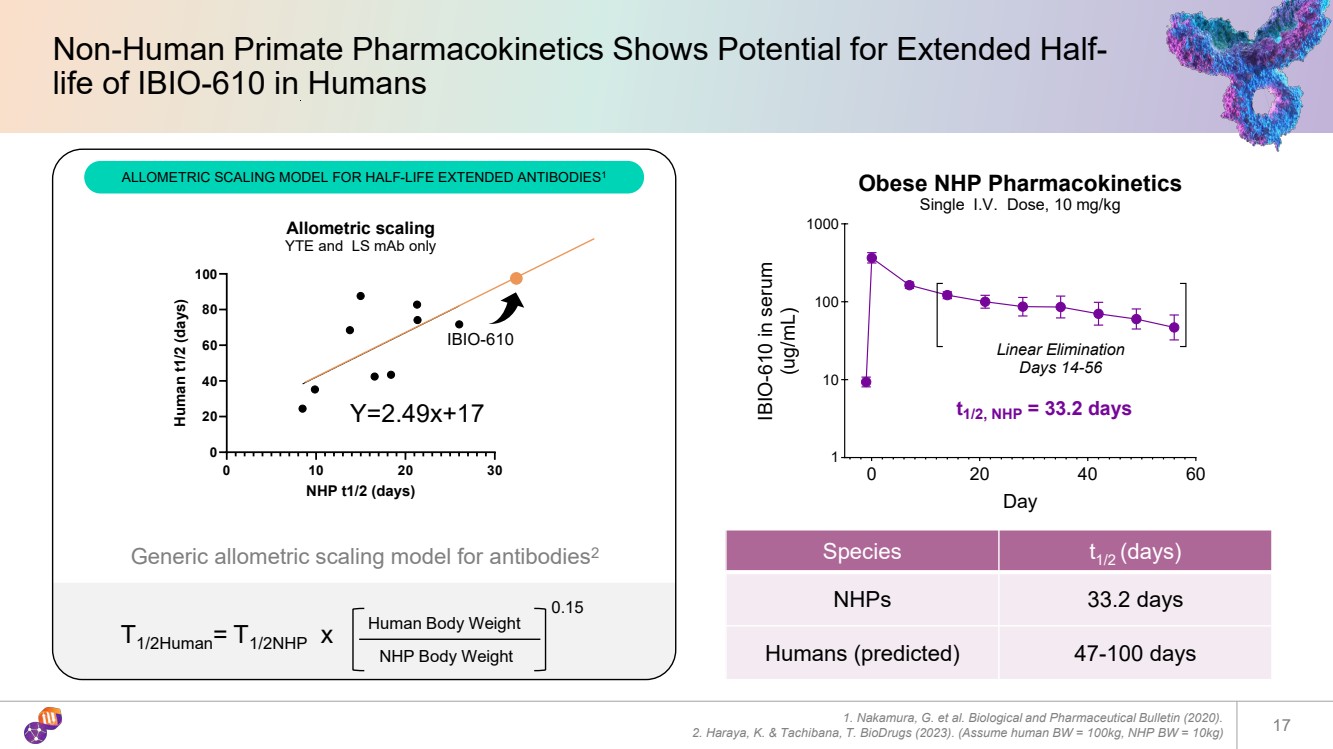
| 17 Non-Human Primate Pharmacokinetics Shows Potential for Extended Half-life of IBIO-610 in Humans 0 20 40 60 1 10 100 1000 Obese NHP Pharmacokinetics Single I.V. Dose, 10 mg/kg Day IBIO-610 in serum (ug/mL) Linear Elimination Days 14-56 t Y=2.49x+17 1/2, NHP = 33.2 days 0 10 20 30 0 20 40 60 80 100 Allometric scaling YTE and LS mAb only NHP t1/2 (days) Human t1/2 (days) Generic allometric scaling model for antibodies2 T1/2Human= T1/2NHP x Human Body Weight NHP Body Weight 0.15 IBIO-610 1. Nakamura, G. et al. Biological and Pharmaceutical Bulletin (2020). 2. Haraya, K. & Tachibana, T. BioDrugs (2023). (Assume human BW = 100kg, NHP BW = 10kg) Species t1/2 (days) NHPs 33.2 days Humans (predicted) 47-100 days ALLOMETRIC SCALING MODEL FOR HALF-LIFE EXTENDED ANTIBODIES1 |
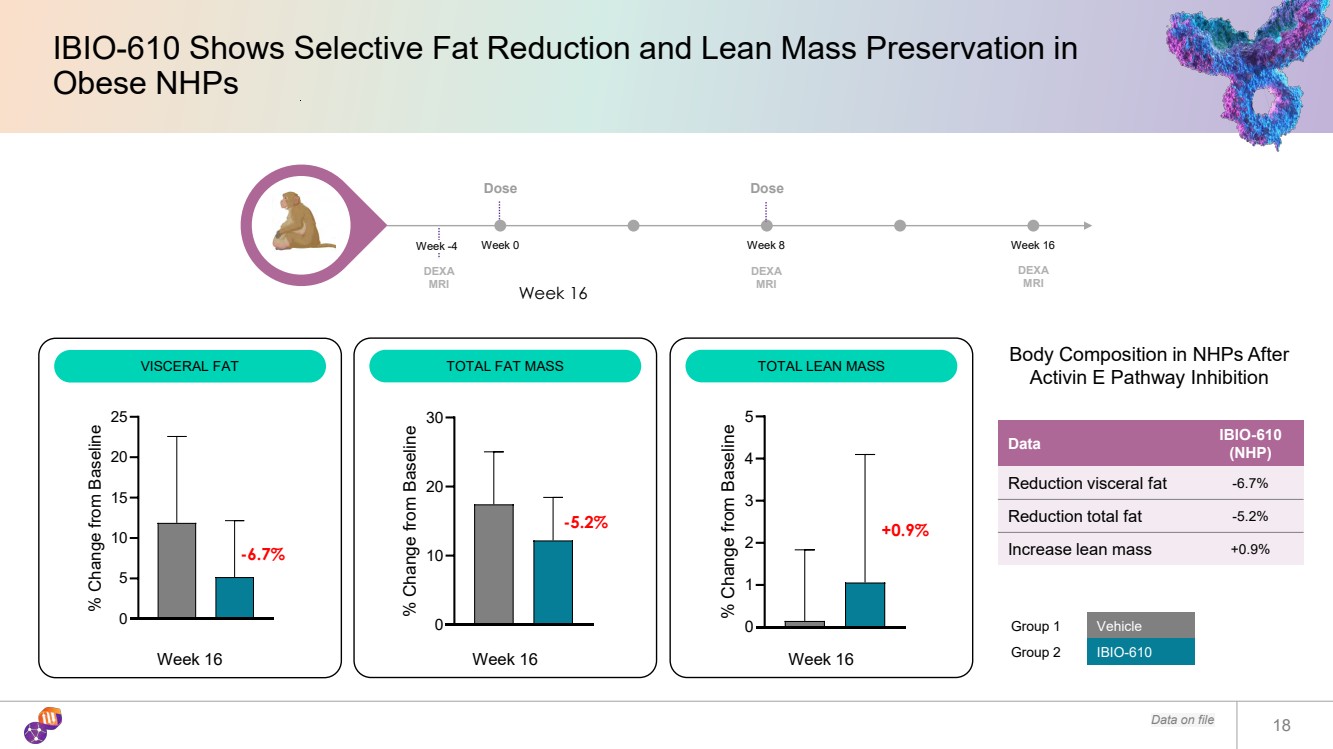
| Data on file 18 IBIO-610 Shows Selective Fat Reduction and Lean Mass Preservation in Obese NHPs Group 1 Vehicle Group 2 IBIO-610 0 5 10 15 20 25 % Change from Baseline 0 10 20 30 % Change from Baseline 0 1 2 3 4 5% Change from Baseline Week 16 -6.7% -5.2% +0.9% Week 16 Data IBIO-610 (NHP) Reduction visceral fat -6.7% Reduction total fat -5.2% Increase lean mass +0.9% Body Composition in NHPs After Activin E Pathway Inhibition Dose Week 0 Week 8 Week 16 DEXA MRI DEXA MRI Dose DEXA MRI Week -4 VISCERAL FAT Week 16 TOTAL FAT MASS Week 16 TOTAL LEAN MASS |
| The Next Wave of iBio Innovation Early Preclinical Programs |
| 20 Combined Myostatin and Activin A Antagonism Precision Bispecific Approach to Address Root Drivers of PH-HFpEF* Binding of Myostatin and Activin A to cells leads to muscle atrophy Simultaneous blocking of Myostatin and Activin A leads to muscle growth Targeting the Core Biology of Disease Across Multiple Organs • Our bispecific antibody is designed to selectively neutralize what are believed to be key pathological ligands: • Activin A → cardiac fibrosis and vascular remodeling1 • Myostatin/GDF11 → skeletal muscle dysfunction and exercise intolerance2,3 • Together, these pathways address two major biological drivers of PH-HFpEF. Selective Modulation Rather Than Broad Pathway Blockade4 • Our bispecific antibody is constructed from first principles to achieve intentional selectivity • Intentionally spares related signaling molecules maintaining physiologic balance • Focused on disease-driving biology Platform-Enabled Precision Engineering • Mammalian display platform allows rapid generation of highly selective multispecific antibodies • Design strategy enabled by iBio’s experience in the TGF-β superfamily Strategic Value • A differentiated, multi-organ therapeutic approach in a clinically validated pathway with strong partnering potential. 1. Roh, J., et al. Sci Transl Med (2020) 2. Abati, E., et al. Cell Mol Life Sci (2022) 3. Zimmers, T.. et al. Basic Res Cardiol (2017) 4. Lan, Z., Lv, Z., Zuo, W. & Xiao, Y. From bench to bedside: The promise of sotatercept in hematologic disorders. Biomedicine & Pharmacotherapy 165, 115239 (2023) * Pulmonary Hypertension (PH) in Heart Failure With Preserved Ejection Fraction (HFpEF) |
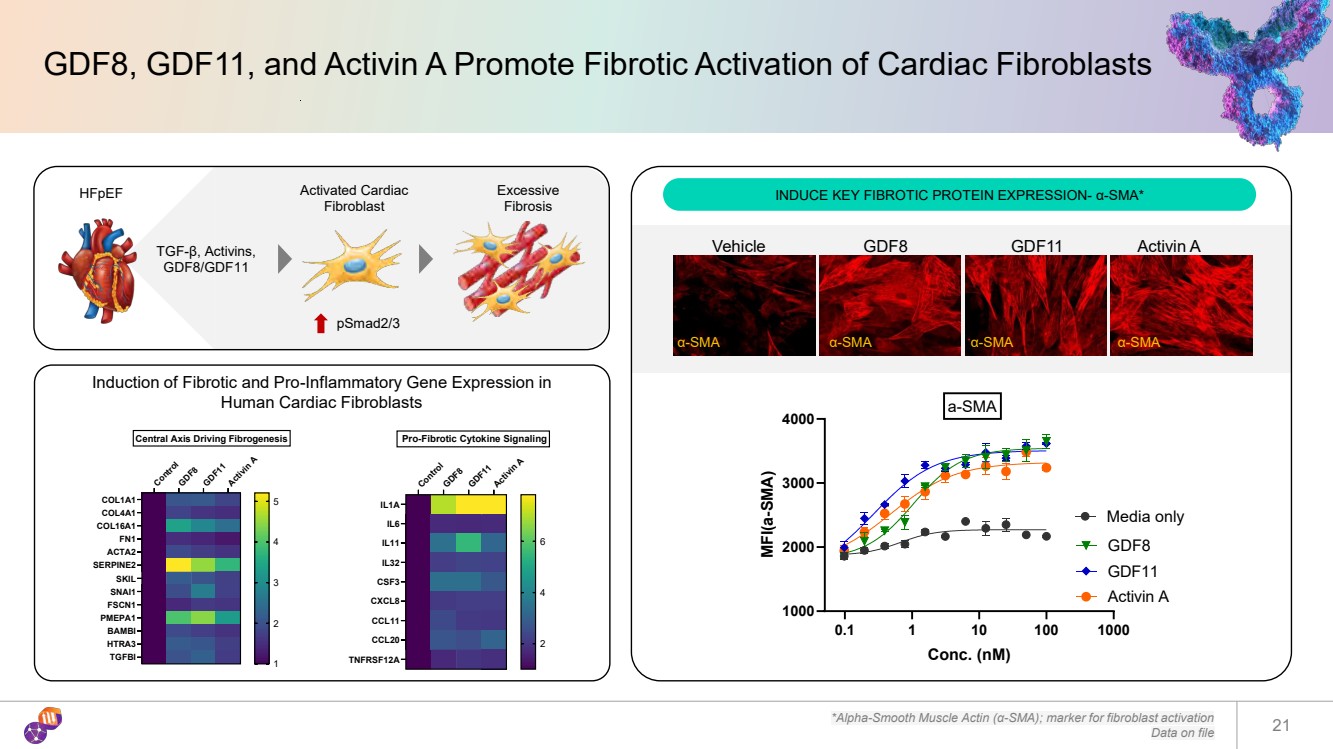
| Induction of Fibrotic and Pro-Inflammatory Gene Expression in Human Cardiac Fibroblasts *Alpha-Smooth Muscle Actin (α-SMA); marker for fibroblast activation Data on file 21 Control GDF8GDF11Activin A COL1A1 COL4A1 COL16A1 FN1 ACTA2 SERPINE2 SKIL SNAI1 FSCN1 PMEPA1 BAMBI HTRA3 TGFBI Central Axis Driving Fibrogenesis 1 2 3 4 5 Control GDF8GDF11Activin A IL1A IL6 IL11 IL32 CSF3 CXCL8 CCL11 CCL20 TNFRSF12A Pro-Fibrotic Cytokine Signaling 2 4 6 α-SMA α-SMA α-SMA α-SMA Vehicle GDF8 GDF11 Activin A 0.1 1 10 100 1000 1000 2000 3000 4000 Conc. (nM) MFI(a-SMA) Media only GDF8 GDF11 Activin A a-SMA GDF8, GDF11, and Activin A Promote Fibrotic Activation of Cardiac Fibroblasts HFpEF INDUCE KEY FIBROTIC PROTEIN EXPRESSION- α-SMA* TGF-β, Activins, GDF8/GDF11 Activated Cardiac Fibroblast Excessive Fibrosis pSmad2/3 |
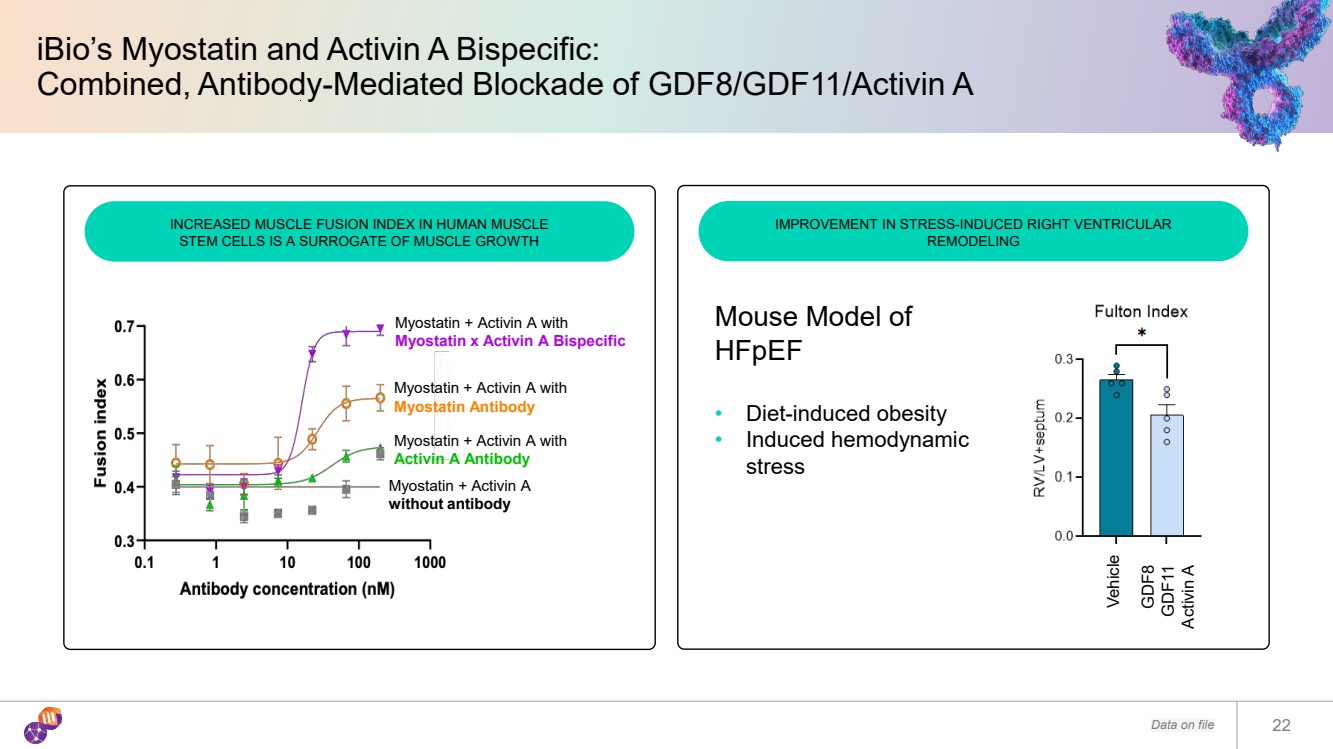
| iBio’s Myostatin and Activin A Bispecific: Combined, Antibody-Mediated Blockade of GDF8/GDF11/Activin A Data on file 22 INCREASED MUSCLE FUSION INDEX IN HUMAN MUSCLE STEM CELLS IS A SURROGATE OF MUSCLE GROWTH IMPROVEMENT IN STRESS-INDUCED RIGHT VENTRICULAR REMODELING Myostatin + Activin A without antibody Myostatin + Activin A with Activin A Antibody Myostatin + Activin A with Myostatin Antibody Myostatin + Activin A with Myostatin x Activin A Bispecific Vehicle GDF8 GDF11 Activin A Mouse Model of HFpEF • Diet-induced obesity • Induced hemodynamic stress |
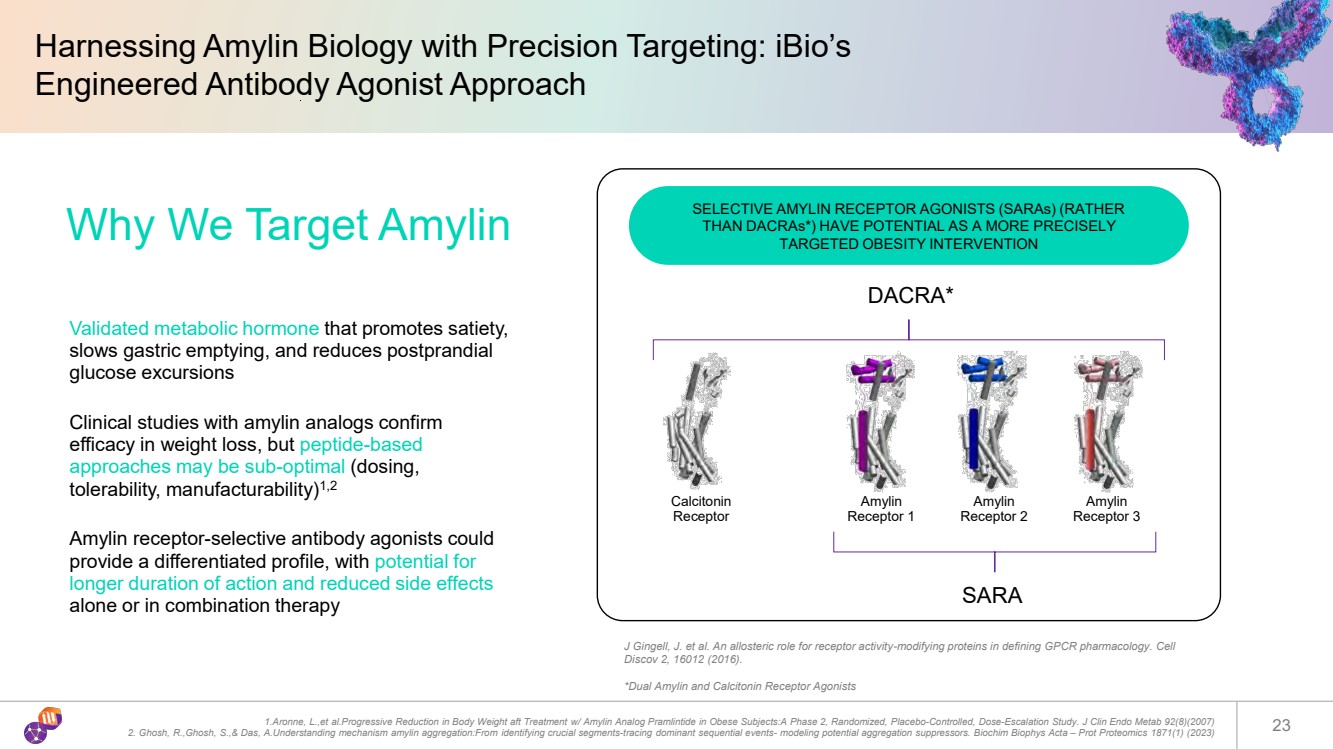
| Harnessing Amylin Biology with Precision Targeting: iBio’s Engineered Antibody Agonist Approach 1.Aronne, L.,et al.Progressive Reduction in Body Weight aft Treatment w/ Amylin Analog Pramlintide in Obese Subjects:A Phase 2, Randomized, Placebo-Controlled, Dose-Escalation Study. J Clin Endo Metab 92(8)(2007) 2. Ghosh, R.,Ghosh, S.,& Das, A.Understanding mechanism amylin aggregation:From identifying crucial segments-tracing dominant sequential events- modeling potential aggregation suppressors. Biochim Biophys Acta – Prot Proteomics 1871(1) (2023) 23 Why We Target Amylin Validated metabolic hormone that promotes satiety, slows gastric emptying, and reduces postprandial glucose excursions Clinical studies with amylin analogs confirm efficacy in weight loss, but peptide-based approaches may be sub-optimal (dosing, tolerability, manufacturability)1,2 Amylin receptor-selective antibody agonists could provide a differentiated profile, with potential for longer duration of action and reduced side effects alone or in combination therapy DACRA* J Gingell, J. et al. An allosteric role for receptor activity-modifying proteins in defining GPCR pharmacology. Cell Discov 2, 16012 (2016). *Dual Amylin and Calcitonin Receptor Agonists Calcitonin Receptor Amylin Receptor 1 Amylin Receptor 2 Amylin Receptor 3 SARA SELECTIVE AMYLIN RECEPTOR AGONISTS (SARAs) (RATHER THAN DACRAs*) HAVE POTENTIAL AS A MORE PRECISELY TARGETED OBESITY INTERVENTION |
| IBIO-600 Long-Acting Myostatin Antibody |
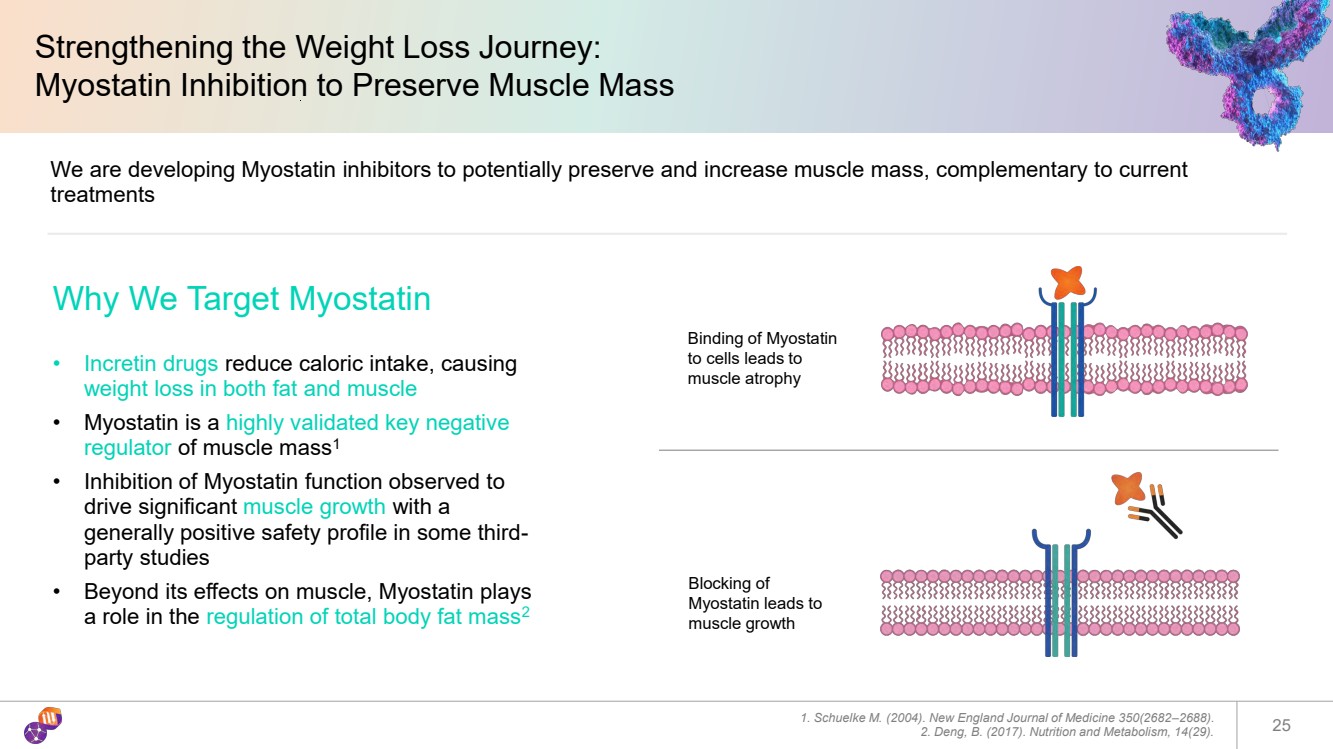
| Strengthening the Weight Loss Journey: Myostatin Inhibition to Preserve Muscle Mass 1. Schuelke M. (2004). New England Journal of Medicine 350(2682–2688). 2. Deng, B. (2017). Nutrition and Metabolism, 14(29). 25 We are developing Myostatin inhibitors to potentially preserve and increase muscle mass, complementary to current treatments Why We Target Myostatin • Incretin drugs reduce caloric intake, causing weight loss in both fat and muscle • Myostatin is a highly validated key negative regulator of muscle mass1 • Inhibition of Myostatin function observed to drive significant muscle growth with a generally positive safety profile in some third-party studies • Beyond its effects on muscle, Myostatin plays a role in the regulation of total body fat mass2 Binding of Myostatin to cells leads to muscle atrophy Blocking of Myostatin leads to muscle growth |
| IBIO-600: A Differentiated Long Acting Anti-Myostatin Program 26 IMPROVED PHARMACOKINETICS Potential best-in-class PK based on allometric scaling and dosing regimen suggests 2-4x improved PK over competitors DUAL MECHANISM Dual myostatin and GDF11 blockade has potential for improved lean mass preservation and fat mass reduction ENHANCED MANUFACTURABILITY Optimized for high expression and stability to enable efficient manufacturing process COFORMULATION OPTIONALITY High formulation concentration to lower injection volume CONVENIENCE Potential administration potentially as infrequent as twice a year |
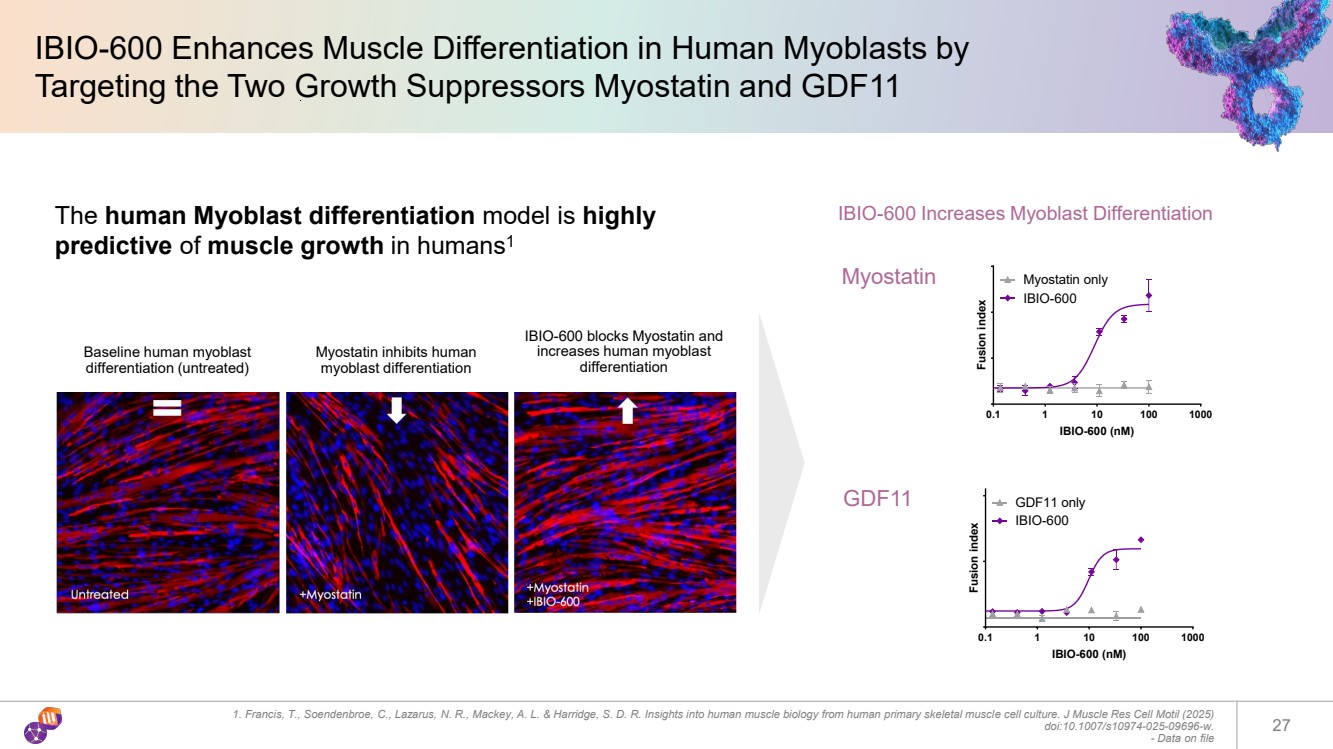
| IBIO-600 Enhances Muscle Differentiation in Human Myoblasts by Targeting the Two Growth Suppressors Myostatin and GDF11 1. Francis, T., Soendenbroe, C., Lazarus, N. R., Mackey, A. L. & Harridge, S. D. R. Insights into human muscle biology from human primary skeletal muscle cell culture. J Muscle Res Cell Motil (2025) doi:10.1007/s10974-025-09696-w. - Data on file 27 IBIO-600 Increases Myoblast Differentiation Baseline human myoblast differentiation (untreated) Myostatin inhibits human myoblast differentiation IBIO-600 blocks Myostatin and increases human myoblast differentiation The human Myoblast differentiation model is highly predictive of muscle growth in humans1 0.1 1 10 100 1000 IBIO-600 (nM) Fusion index Myostatin only IBIO-600 0.1 1 10 100 1000 IBIO-600 (nM) Fusion index GDF11 only IBIO-600 Myostatin GDF11 |
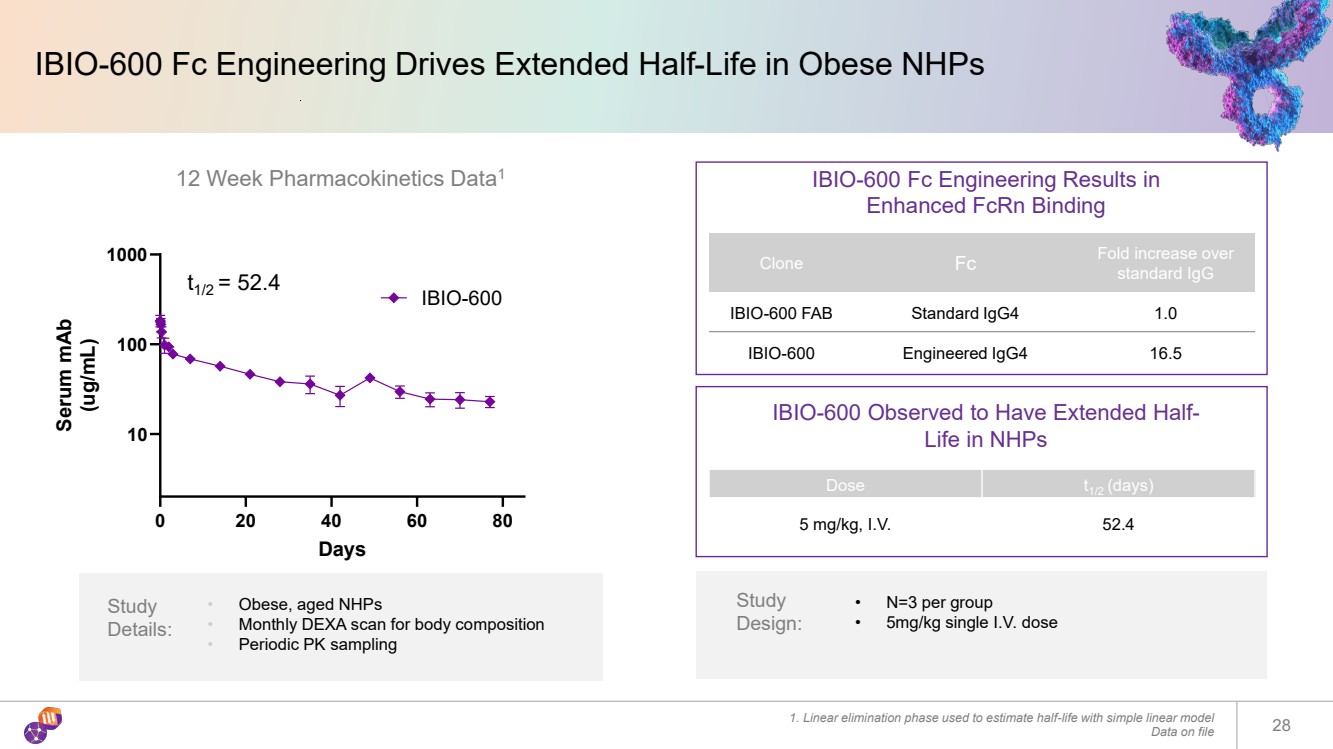
| IBIO-600 Fc Engineering Drives Extended Half-Life in Obese NHPs 1. Linear elimination phase used to estimate half-life with simple linear model Data on file 28 Dose t1/2 (days) 5 mg/kg, I.V. 52.4 IBIO-600 Fc Engineering Results in Enhanced FcRn Binding Clone Fc Fold increase over standard IgG IBIO-600 FAB Standard IgG4 1.0 IBIO-600 Engineered IgG4 16.5 IBIO-600 Observed to Have Extended Half-Life in NHPs 12 Week Pharmacokinetics Data1 0 20 40 60 80 10 100 1000 Days Serum mAb (ug/mL) IBIO-600 Study Details: • Obese, aged NHPs • Monthly DEXA scan for body composition • Periodic PK sampling Study Design: • N=3 per group • 5mg/kg single I.V. dose t1/2 = 52.4 |
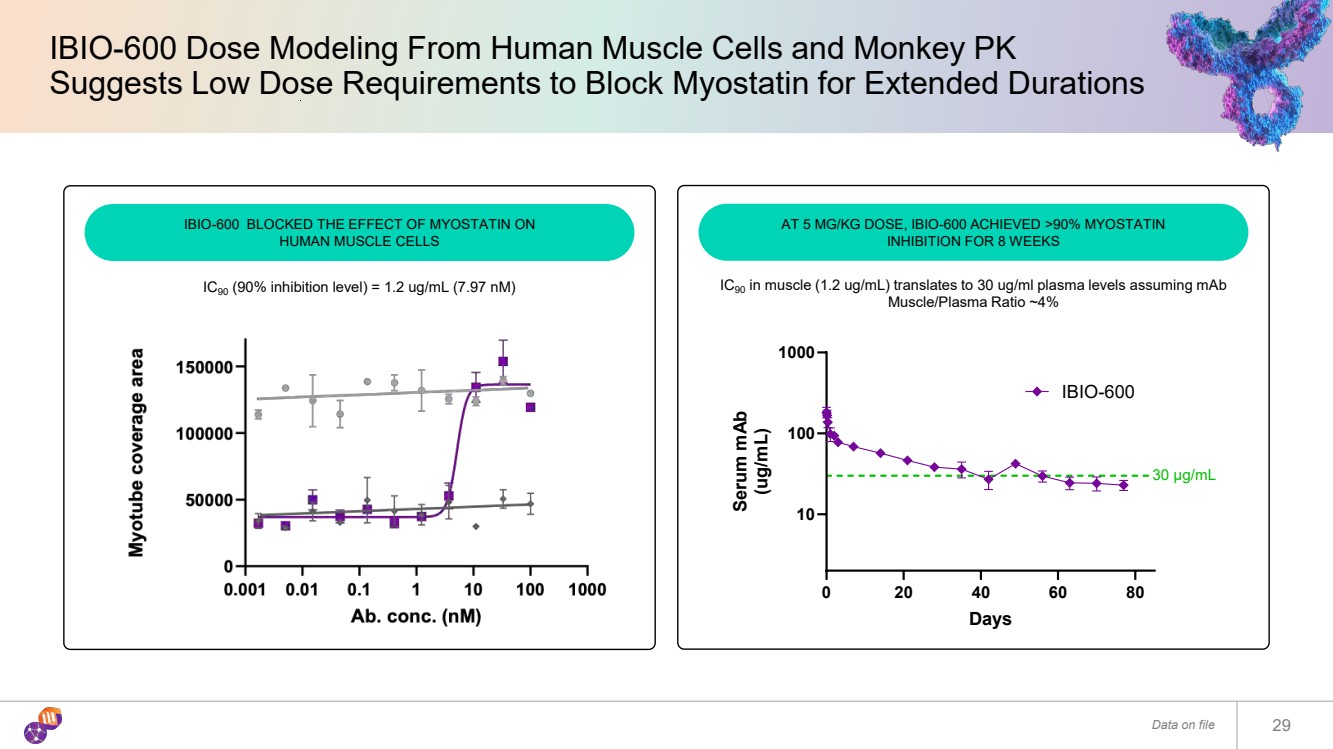
| 29 IBIO-600 Dose Modeling From Human Muscle Cells and Monkey PK Suggests Low Dose Requirements to Block Myostatin for Extended Durations Data on file IBIO-600 BLOCKED THE EFFECT OF MYOSTATIN ON HUMAN MUSCLE CELLS AT 5 MG/KG DOSE, IBIO-600 ACHIEVED >90% MYOSTATIN INHIBITION FOR 8 WEEKS IC90 (90% inhibition level) = 1.2 ug/mL (7.97 nM) 0 20 40 60 80 10 100 1000 Days Serum mAb (ug/mL) IBIO-600 30 μg/mL IC90 in muscle (1.2 ug/mL) translates to 30 ug/ml plasma levels assuming mAb Muscle/Plasma Ratio ~4% |
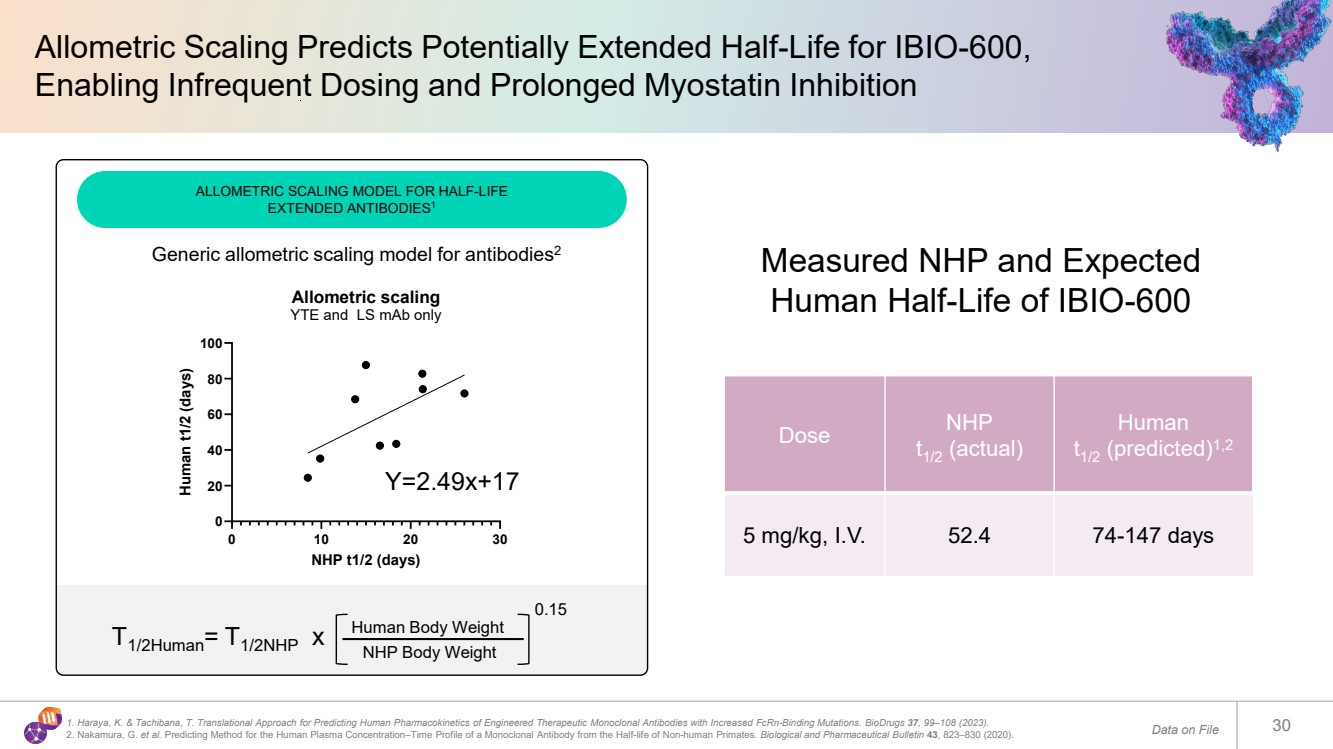
| Allometric Scaling Predicts Potentially Extended Half-Life for IBIO-600, Enabling Infrequent Dosing and Prolonged Myostatin Inhibition Data on File 30 Dose NHP t1/2 (actual) Human t1/2 (predicted)1,2 5 mg/kg, I.V. 52.4 74-147 days Measured NHP and Expected Human Half-Life of IBIO-600 1. Haraya, K. & Tachibana, T. Translational Approach for Predicting Human Pharmacokinetics of Engineered Therapeutic Monoclonal Antibodies with Increased FcRn-Binding Mutations. BioDrugs 37, 99–108 (2023). 2. Nakamura, G. et al. Predicting Method for the Human Plasma Concentration–Time Profile of a Monoclonal Antibody from the Half-life of Non-human Primates. Biological and Pharmaceutical Bulletin 43, 823–830 (2020). ALLOMETRIC SCALING MODEL FOR HALF-LIFE EXTENDED ANTIBODIES1 Y=2.49x+17 0 10 20 30 0 20 40 60 80 100 Allometric scaling YTE and LS mAb only NHP t1/2 (days) Human t1/2 (days) Generic allometric scaling model for antibodies2 T1/2Human= T1/2NHP x Human Body Weight NHP Body Weight 0.15 |
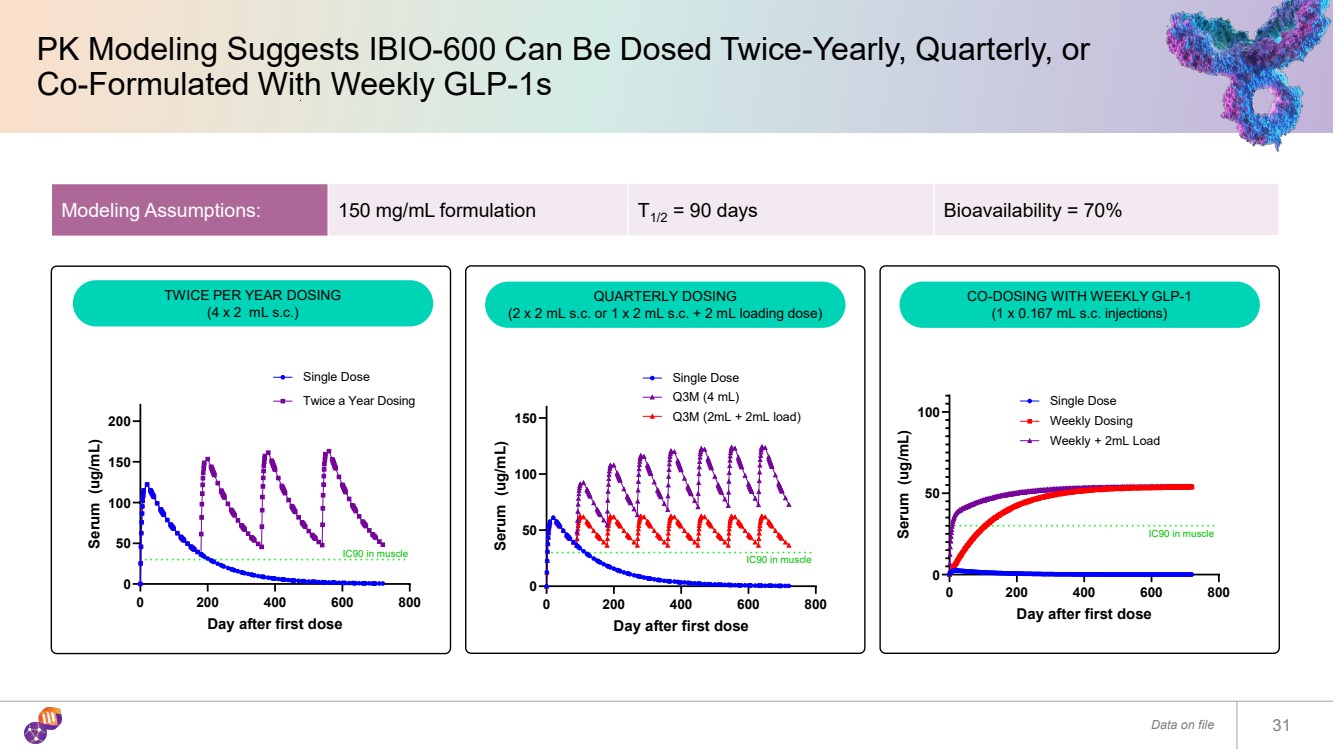
| PK Modeling Suggests IBIO-600 Can Be Dosed Twice-Yearly, Quarterly, or Co-Formulated With Weekly GLP-1s Data on file 31 0 200 400 600 800 0 50 100 150 200 Day after first dose Serum (ug/mL) Single Dose Twice a Year Dosing IC90 in muscle 0 200 400 600 800 0 50 100 Day after first dose Serum (ug/mL) Single Dose Weekly Dosing IC90 in muscle Weekly + 2mL Load Modeling Assumptions: 150 mg/mL formulation T1/2 = 90 days Bioavailability = 70% 0 200 400 600 800 0 50 100 150 Day after first dose Serum (ug/mL) Single Dose Q3M (4 mL) IC90 in muscle Q3M (2mL + 2mL load) TWICE PER YEAR DOSING (4 x 2 mL s.c.) QUARTERLY DOSING (2 x 2 mL s.c. or 1 x 2 mL s.c. + 2 mL loading dose) CO-DOSING WITH WEEKLY GLP-1 (1 x 0.167 mL s.c. injections) |
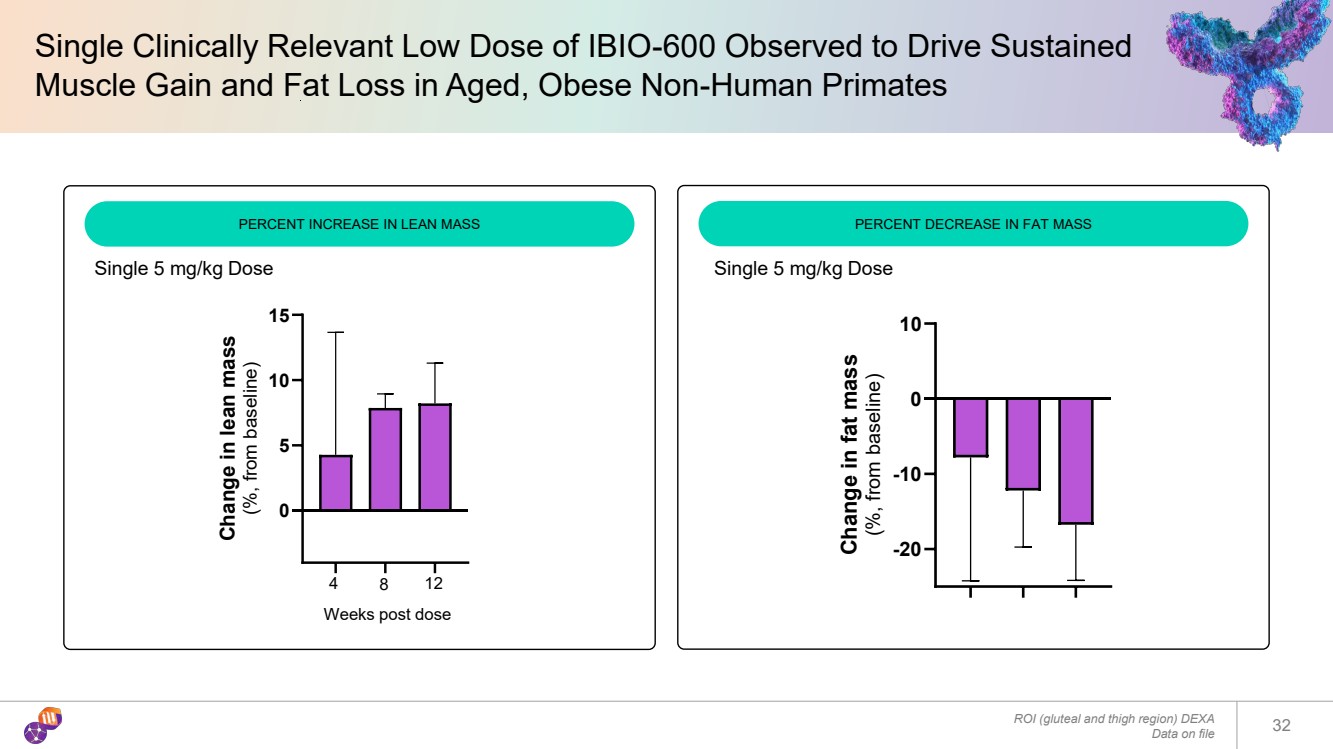
| Single Clinically Relevant Low Dose of IBIO-600 Observed to Drive Sustained Muscle Gain and Fat Loss in Aged, Obese Non-Human Primates 32 ROI (gluteal and thigh region) DEXA Data on file PERCENT INCREASE IN LEAN MASS PERCENT DECREASE IN FAT MASS Weeks post dose 0 5 10 15 Change in lean mass (%, from baseline) 4 8 12 -20 -10 0 10 Change in fat mass (%, from baseline) Single 5 mg/kg Dose Single 5 mg/kg Dose |
| Technology Stack |
| Toward Any Epitope on Any Drug Target AI Epitope Engineering and Antibody Optimization Engines unlock challenging target classes 34 • Multi-layer technology platform addresses multiple challenges in Ab discovery • Patented Epitope Steering technology • Single-step Ab StableHu x Mammalian Display • Masked (ShieldTx®) Antibodies • T-cell engager panel (EngageTx ) iBio’s Discovery Engine iBio’s Proprietary AI Technology Platform We use our Tech Stack to generate new IP against hard-to-drug targets – from idea to Development Candidate in 7 months • Selectively targets functional epitopes • Epitopes with complex modes of action • Unlocks novel target classes • Accelerates discovery of Ab against validated targets AI-guided precision hits that are epitope class agnostic • Gen AI creates mammalian display libraries with phage-like diversity • Single-shot multidimensional lead optimization • Compatible with multi-specific antibody formats • Antibody format agnostic Generative AI meets mammalian display: Ab optimization in 3 weeks |
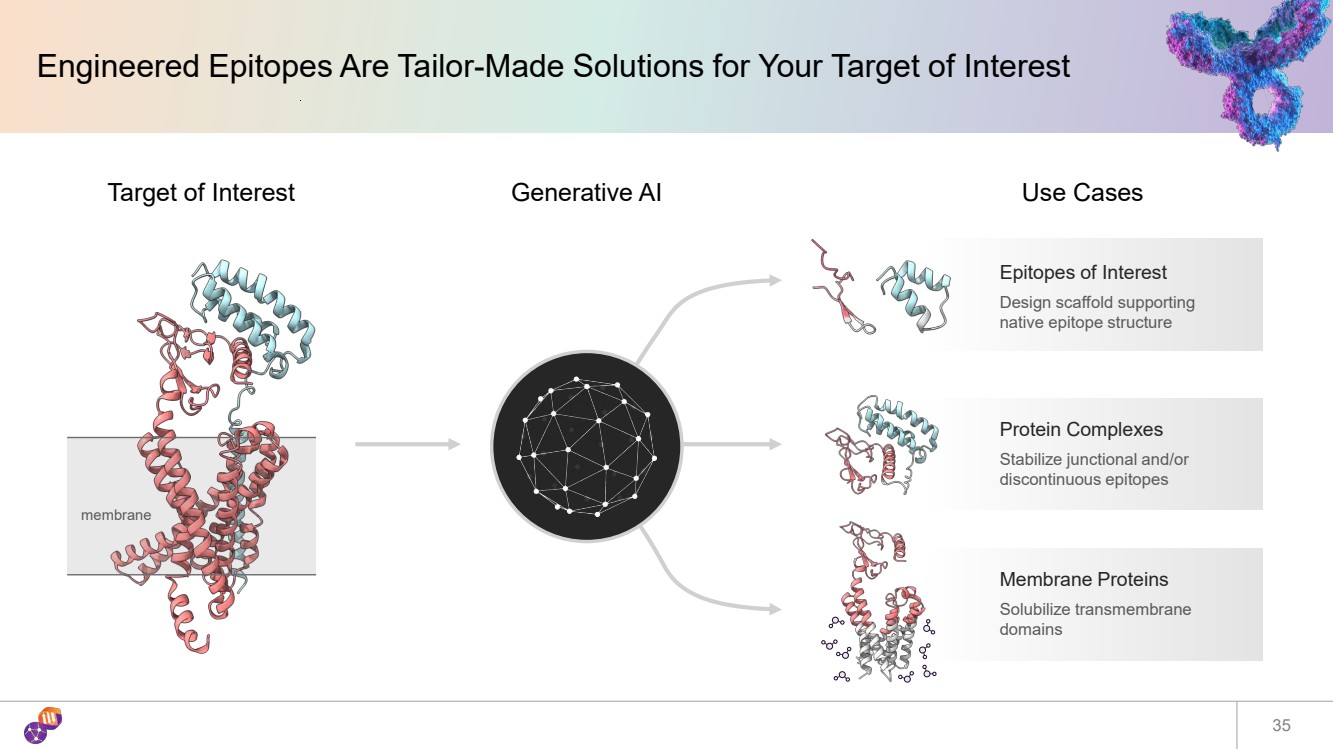
| Engineered Epitopes Are Tailor-Made Solutions for Your Target of Interest 35 Generative AI Protein Complexes Stabilize junctional and/or discontinuous epitopes Target of Interest Epitopes of Interest Design scaffold supporting native epitope structure Membrane Proteins Solubilize transmembrane domains Use Cases membrane |
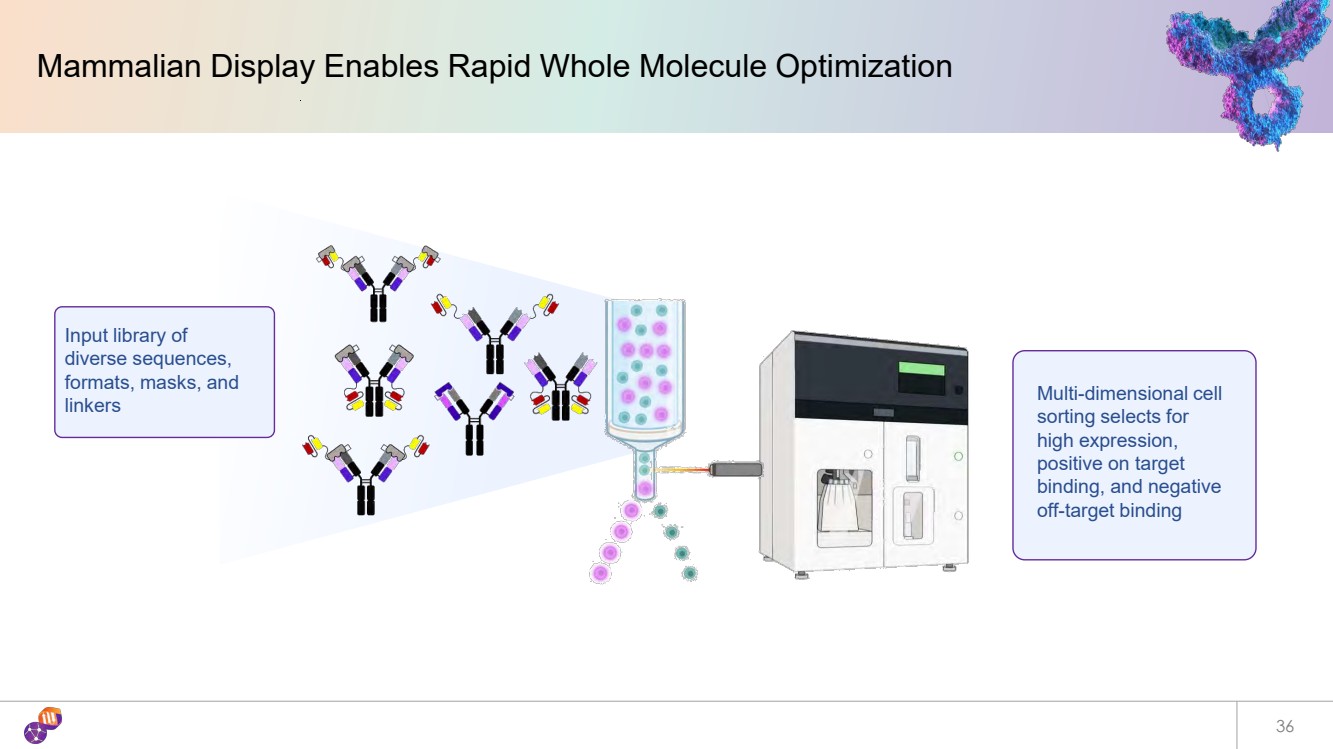
| 36 Mammalian Display Enables Rapid Whole Molecule Optimization Input library of diverse sequences, formats, masks, and linkers Multi-dimensional cell sorting selects for high expression, positive on target binding, and negative off-target binding |
| Corporate Summary |
| A Leadership Team with Deep Industry Experience 38 Martin Brenner, DVM, Ph.D. CEO & CSO Felipe Duran CFO Marc Banjak CLO |
| Executive Summary Corporate Highlights Differentiated Pipeline Aiming to Solve for the Challenges of today’s GLP1’s ▪ Focus on increased quality of weight loss (IBIO-610, Myostatin x Activin A) ▪ Developability (IBIO-610) Patented AI-Driven Discovery Tech Stack ▪ Advance a highly developable pre-clinical pipeline ▪ Designed to solve high-value, hard-to-drug targets Financial Highlights 39 ▪ $52.7M in cash, cash equivalents and debt securities1 as of December 31, 2025 ▪ Net proceeds of ~$24.4M from PIPE financing in January 2026 ▪ ~34.5M shares outstanding as of February 24, 2026 ▪ ~89.0M shares issuable upon exercise of pre-funded warrants outstanding at a weighted average exercise price of $0.001 per share ▪ Cash runway extends into Q3 FY 2028 1. We estimate our cash, cash equivalents, and debt securities to be approximately $52.7 million as of December 31, 2026. This is a preliminary, estimated, and unaudited financial result. Actual results may differ from this estimate. |